Properly measuring our pH is essential for obtaining reliable results that help us evaluate our progress in terms of health and its relationship with alkalinity. Discover these tips for measuring your pH.
Tips for Measuring Your pH
By now, we all understand the important role the pH balance of our body plays and how its variations affect our physical, mental, and emotional health. Therefore, it is crucial to learn how to properly measure our pH, as it serves as a guide to evaluate our health improvements as we adopt an alkaline diet and lifestyle.
The pH scale is logarithmic.
Before explaining how to properly measure our pH, it’s important to clarify that the pH scale ranges from 1 to 14, with 7 being neutral. Anything above 7 is considered alkaline, and anything below is acidic. The most important thing to understand about this scale, which helps us grasp the real impact of a “small” pH variation, is that it is logarithmic.
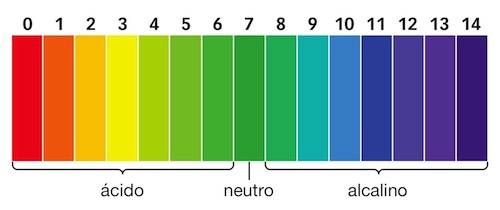
This means that the values are multiplied by 10 with each unit. For example, a pH of 6 is 10 times more acidic than a pH of 7, and a pH of 5 is 100 times more acidic than a pH of 7. Similarly, a pH of 8 is 10 times more alkaline than a pH of 7, and a pH of 9 is 100 times more alkaline than a pH of 7. Understanding this, can you imagine the improvement of going from a pH of 6 to 7? A quantum leap for our health!
What is the Difference Between Measuring pH in Saliva or Urine?
Measuring pH in saliva primarily indicates the state of your mineral reserves, while measuring pH in urine reflects how acidic or alkaline your internal fluids are.
Additionally, urine pH results tend to be more reliable, as saliva pH can vary more easily due to food, beverages, speaking, exhaling, and other factors.
How to Perform a More Accurate pH Measurement
-
- Ideally, measure saliva pH at least an hour before or after meals.
- When testing first thing in the morning, it’s important to brush your teeth and tongue thoroughly beforehand to remove toxins accumulated overnight. During sleep, the mouth is closed, preventing the expulsion of toxins through breath, leaving them trapped on mucous membranes, teeth, and the tongue.
- Avoid keeping the pH strip in your mouth too long. Hold it just long enough to soak it adequately with saliva. Otherwise, you might absorb the strip’s reactive agent, invalidating the test, as the strip will not change color because the reagent has been “consumed.”
Frequency of Use:
-
-
- Measure at least twice a day.
- The first measurement should be with your first urine of the morning. Overnight, the body retains fluids for several hours and performs its detox processes, so the result will show a more acidic pH due to the concentration of toxins.
- After a few hours, repeat the test to get a more balanced result.
- When measuring urine pH, let the urine flow for a few seconds before soaking the pH strip, rather than using the first stream.
- Measure at least twice a day.
-
Long-Term Measurement:
-
- Whether measuring saliva or urine, it is important to take daily measurements over a relatively long period (1–2 weeks). This is because daily factors like diet, sleep, and water intake vary.
- Calculating an average of the results obtained is the best way to get an accurate understanding of how acidic or alkaline your body is.
Systems for Measuring Your pH
pH Test Strips: The Most Practical Tool for Measuring Your pH
pH test paper strips are the most common and widely used tool for measuring pH, not only because they are affordable but also due to their convenience and practicality.
How They Work
These strips are coated with substances that help measure the concentration of hydrogen ions in certain liquid solutions or fluids. To perform a measurement correctly, follow these two simple steps:
- Contact the test strip with the liquid to be measured for a few seconds, just enough time for it to soak with the substance.
- Compare the resulting color with the color scale provided with the pH test strips.
The usability of these strips varies depending on the reagents they contain, as different liquids require different reagents. For this reason, it’s important to check with the manufacturer or distributor before purchasing to ensure the strips suit your intended use.
Other Methods
Beyond test strips, there are other systems available for measuring pH:
- pH Meter or Potentiometer:
This is a more sensitive and complex device than pH test strips. Its use is less domestic and more suited to laboratory settings. With a pH meter, you can measure any liquid or fluid. Generally, pH meters include two electrodes: a glass sensor and a reference tube. When immersed in a liquid, the sensor measures the activity of hydrogen ions by generating a small electrical current.
The voltage meter interprets this current and converts it into a pH value, displaying the result on a digital screen.

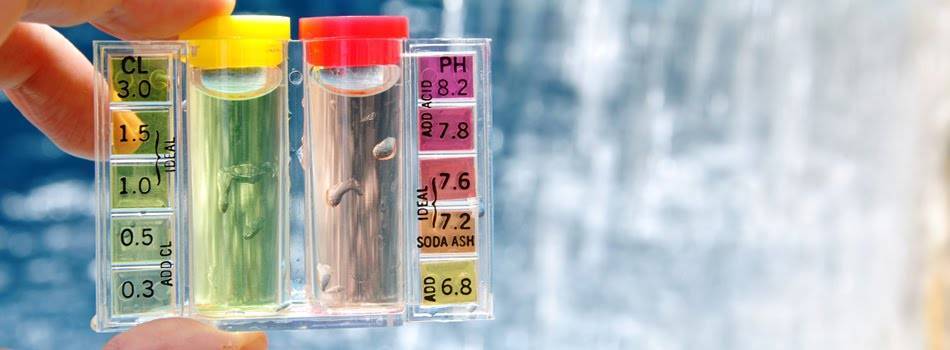
- pH Testing Drops
These are a reactive liquid that changes color when it comes into contact with the water or fluid being tested. The test is very simple. Its function is based on a chemical reaction: a few drops of the reactive liquid are added to the quantity of liquid to be measured. The pH value is determined by comparing the resulting color with a color scale and corresponding values provided by the manufacturer along with the drops.